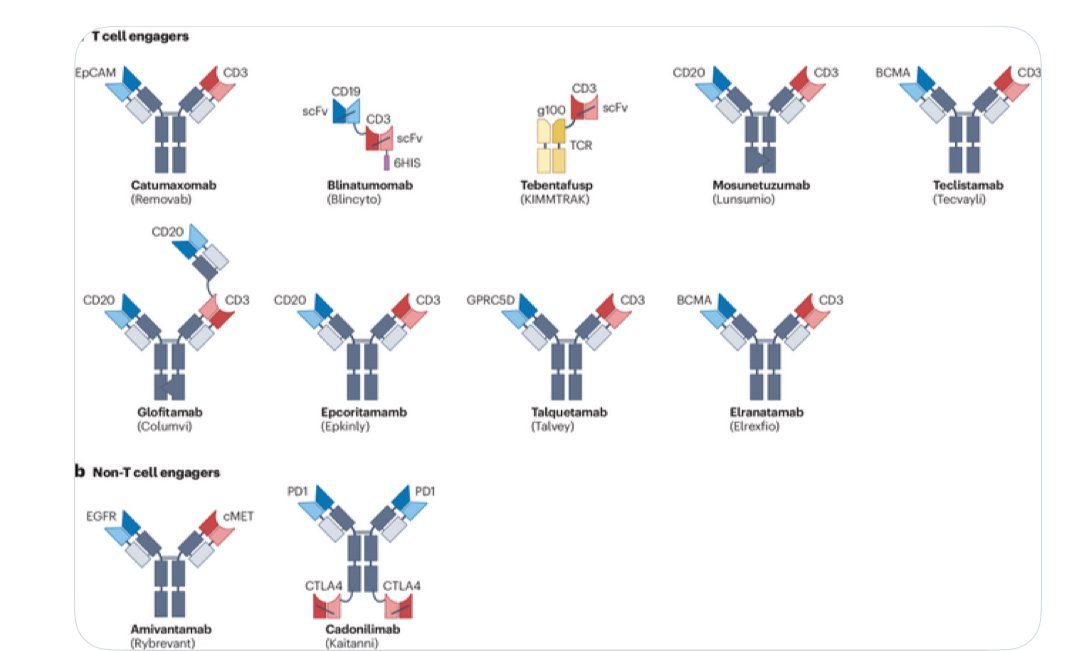
Bispecific antibodies (bsAbs) provide novel mechanisms of action and therapeutic possibilities beyond those achievable with conventional IgG-based antibodies. Their development has accelerated over the past decade, leading to the approval of 14 bsAbs by the end of 2023—11 for oncology indications and 3 for non-oncology uses. These molecules come in various formats, target a wide range of antigens, and mediate anti-tumor activity through diverse molecular mechanisms.
A recent review published in Nature summarizes key advances in bsAbs for cancer therapy, emphasizing agents that are either approved or in clinical development. Highlighted categories include dual modulators of signaling pathways, tumor-targeted receptor agonists, bsAb–drug conjugates, and bsAbs that recruit T cells, natural killer (NK) cells, or other innate immune effectors. The review also explores bispecific checkpoint inhibitors and co-stimulatory molecules.
Finally, the authors discuss next-generation bsAb platforms in early-stage development, such as trispecific antibodies, bsAb prodrugs, tumor-targeting degraders, and bsAbs designed to function as cytokine mimetics.
Klein C, Brinkmann U, Reichert J, Kontermann R. “The present and future of bispecific antibodies for cancer therapy.” Nat Rev Drug Discov. 2024 Mar 6;23(4):237–257. doi:10.1038/s41573-024-00896-6. PMID: 38448606.
SHARE


